 
For a simple cubic lattice, the packing efficiency is 52.4% and the packing efficiency is 68% for a body-centered cubic lattice or bcc. The packing efficiency of both of these is 74% which means 74% of the space is filled. The cubic closed packed or ccp and the hexagonal closed packed or hcp are two efficient lattices when we consider packing. After reaching a yield stress at t x 180, t y 110 ( reduced units ), we observe a sudden increase of the amount of hcp structure as demonstrated in Fig. This unit cell of the lattice is a three-dimensional structure that has one or more atoms and also void spaces irrespective of the packing present. When it is represented as a percentage then the percentage of the space applied by the constituent particles out of the total space present in the structure is called the packing efficiency of the unit cell.Ī lattice is largely made of a number of unit cells in which the lattice point is filled or occupied by a constituent particle. This can be obtained by dividing the volume of the constituent particles filled in the space by the total volume of the cell. The fraction of total space that is filled with the particular cell or structure is called the packing fraction. The space is filled by other constituents or particles. Each atom contacts six atoms in its own layer, three in the layer above, and three in the layer below. In CCP, there are three repeating layers of hexagonally arranged atoms. This structure is also called cubic closest packing (CCP). Even when there is packing in the cell, a certain void is present in it. Atoms in an FCC arrangement are packed as closely together as possible, with atoms occupying 74 of the volume. Unit cell can be defined as a three-dimensional structure that is made of one or more than one atom. In hcp and ccp i.e., Face Centred Cubic StructureĪgain, we draw the face diagonal and as shown in the figure, 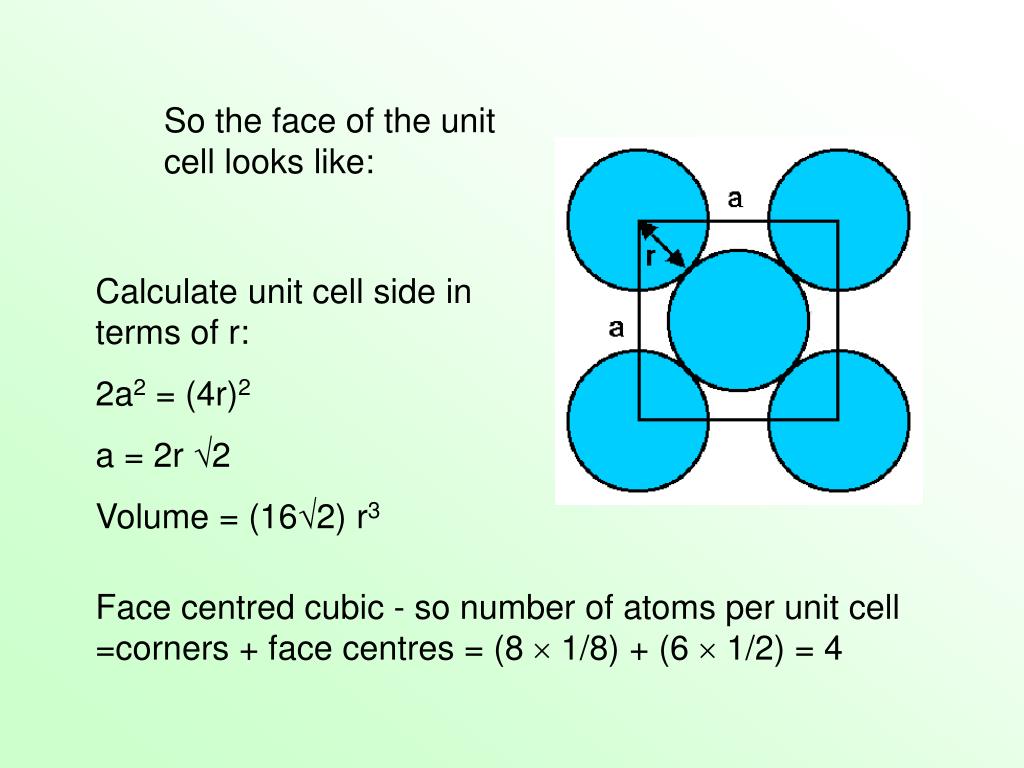
In this case, since atoms are on the corners and an atom is present in the center, we draw a diagonal, and its length (c) can be calculated using Pythagoras theorem.Īnd since radius = 4 X Diagonal (as shown in the figure under Body Centred Cubic Lattice) When this is shown as a percentage i.e., out of the total space, the percentage that is held up by constituent particles is called the Packing Efficiency of a Unit Cell. It can be obtained by dividing the total volume occupied by constituent particles by the cell's total volume. 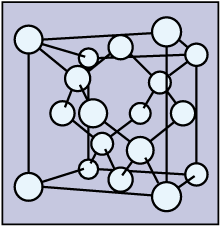
The fraction of total space that is filled with the inherent constituent particles of a particular cell or structure is called the packing fraction. Some void space is always present irrespective of the type of packing the cell has. Mathematically, Atomic Packing Factor (APF):ĪPF \( = \frac\)įor FCC a = 2√2 r where a is side of the cube and r is atomic radius.A Unit Cell may be viewed as a 3-D Structure made up of one or more atoms. The atomic packing factor is defined as the ratio of the volume occupied by the average number of atoms in a unit cell to the volume of the unit cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
